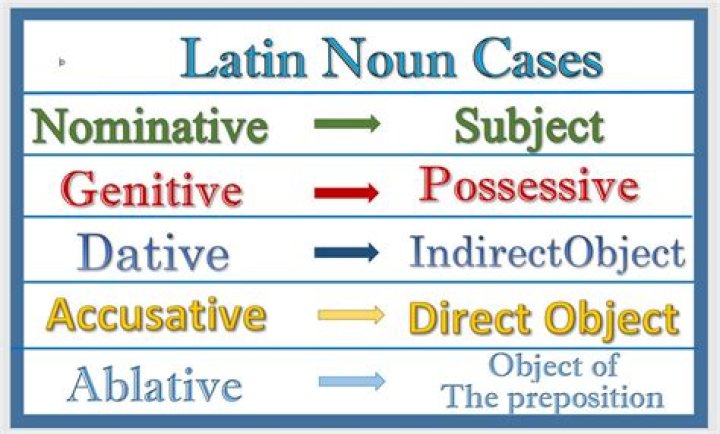
Which defect decreases the density of the crystal?

Robert Guerrero
Updated on May 04, 2026
Also, which defect causes decrease in the density of crystal?
Schottky defect causes decrease in the density of a crystal. In this defect, equal number of cations and anions are missing from their position in the crystal lattice.
Additionally, which defect in ionic solid decreases its density? Answer: In Schottky defect ,the density of an ionic solid is reduced. This defect was discovered by German scientist Schottky.It arises due to missing of same number of cations and anions ions from their positions and thus creating vacancy.. Hence it is also called as Vacancy defect in Ionic solids.
Subsequently, one may also ask, which point defect increases the density of a crystal?
Schottky defect
What is the effect of presence of Schottky defect on the density of a crystal?
Schottky defect : it involves the removal of the equal number of cations and anions( to maintain neutrality of the crystal ) from the ionic crystal. So , as a consequence the density of the crystal decreases.